CMaT Overview
CMaT is a convergence-science-based, strategic international ecosystem for cell therapy manufacturing. CMaT is dedicated to developing new fundamental knowledge; transformative new tools and technologies; integrated next-generation cell-manufacturing platforms; a diverse, inclusive, and well-trained workforce; and best practices, standards, and global thought-leadership. CMaT's 10-year vision is to transform the manufacture of cell-based therapeutics into a scalable, reproducible, lower-cost, and quality-by-design (QbD)-driven engineered system for broad industry and clinical use. This will ultimately improve patient access to new cell-based therapies, significantly impact and improve healthcare, de-risk and advance a nascent industry-sector, add new jobs, and ultimately improve the healthcare economy. CMaT's mission is to be, and be recognized as, a Diverse and Inclusive Innovation Hub for:
• Creating fundamental new knowledge and transformative engineered systems for QbD-driven, robust, lower-cost, and scalable, automation-driven cell manufacturing processes.
• Inventing and translating innovative tools and technologies enabling more affordable, reproducible, and high-quality cell production at various scales.
• Disseminating best practices to all stakeholders throughout the cell manufacturing ecosystem and working with the broader community to enable robust industry standards.
• Training a diverse workforce across the value chain not only on technical aspects but also on policy, ethics, and regulatory issues—including undergraduate students, graduate students, postdoctoral fellows, underrepresented students and teachers from high schools, students with disabilities, and technical and community college students and teachers.
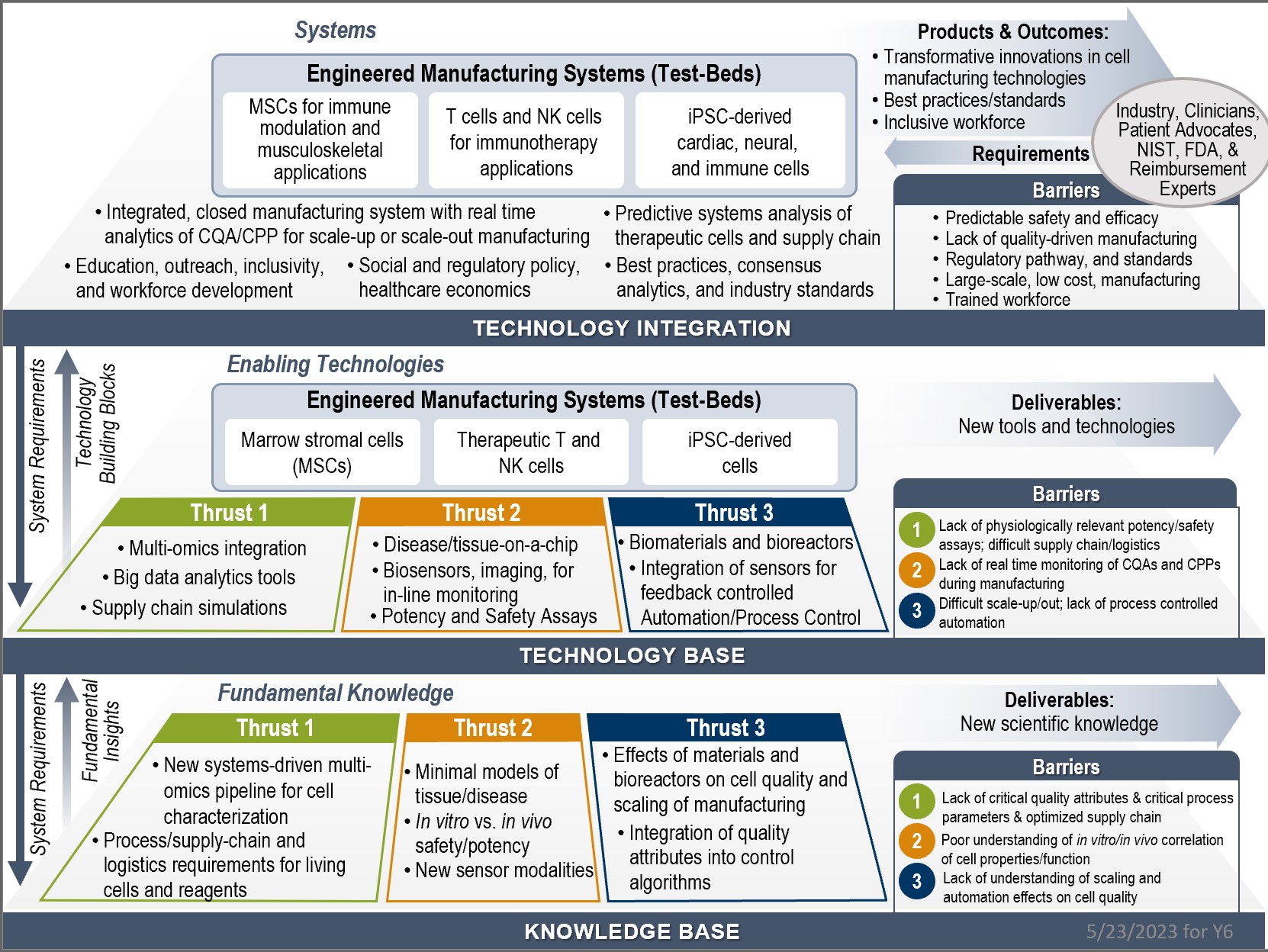
CMaT’s overall research plan and its impact on industry and workforce is shown below. This strategic plan is structured around a broad stakeholder‐driven ecosystem with three inter‐dependent and convergent Thrusts, and three industry‐relevant Test‐Beds (Engineered Systems) that are integrated and cross‐cutting across all three Thrust areas. As shown, CMaT will have tremendous impact in strengthening the nascent industry.
CMaT's Y6 Three-Plane Strategy Chart